Recently I have been making tests with a semi-closed vessel and noticed that the resistance of Kanthal A-1 wires wires treated similarly to previously described would decrease noticeably upon admission of organic compounds like acetone or ethanol.
Acetone in particular would initially combust on the wire, producing a heat burst, but then, as oxygen gas got used up, it decomposed on its surface, causing carbon deposition and presumably hydrogen evolution. I think such hydrogen is what is causing this resistance decrease. At the same time, as the wire gets dark, incandescence becomes dim, perhaps due to the wire acting as a more efficient infrared radiator.

Since the cell is not sealed and air can come in (as well as hydrogen and acetone go out), the wires would eventually acquire back their oxidized color. As this happened, resistance slowly increased back to the starting values.
I tried this with a brand-new bare Kanthal wire and although to a much lesser extent, the effects were still visible (resistance decrease and incandescence dimming).
I then attempted this with a "powered catalyst pellet", which in this case involved embedding the same Kanthal heater in a 75% Fe2O3–25% K2CO3 paste and calcining it to high temperatures to form a hard ceramic pellet similar to actual industrial catalysts, only much larger (approx. 60x6 mm) and with an internal heat source.

Upon admission of about 1.5 ml acetone in the cell (involving temporarily opening it), the previously resistance decrease effect turned out to be much larger, causing a repeatable drop from 5.7 Ohm to about 3.6 Ohm. This drop was repeatable. Notice how turning power off in the second half of the diagram apparently did not affect the trend in resistance.
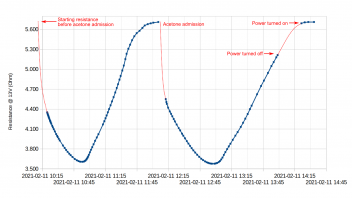
In the process (still keeping the catalyst at a constant 13V with the recently acquired power supply), the powered pellet first got covered in carbon, but eventually, possibly corresponding to the resistance minima in the above graph, the carbon apparently slowly started combusting and eventually mostly disappear. Resistance would then return back to roughly the initial values.


After a few mishaps following such testing, eventually the ceramic portion broke in half, although the wire didn't. After a period at room temperature the material looks red.

It's not clear how much this effect is related to hydrogen absorption, but it's interesting that it could be easily observed with this kind of material.
